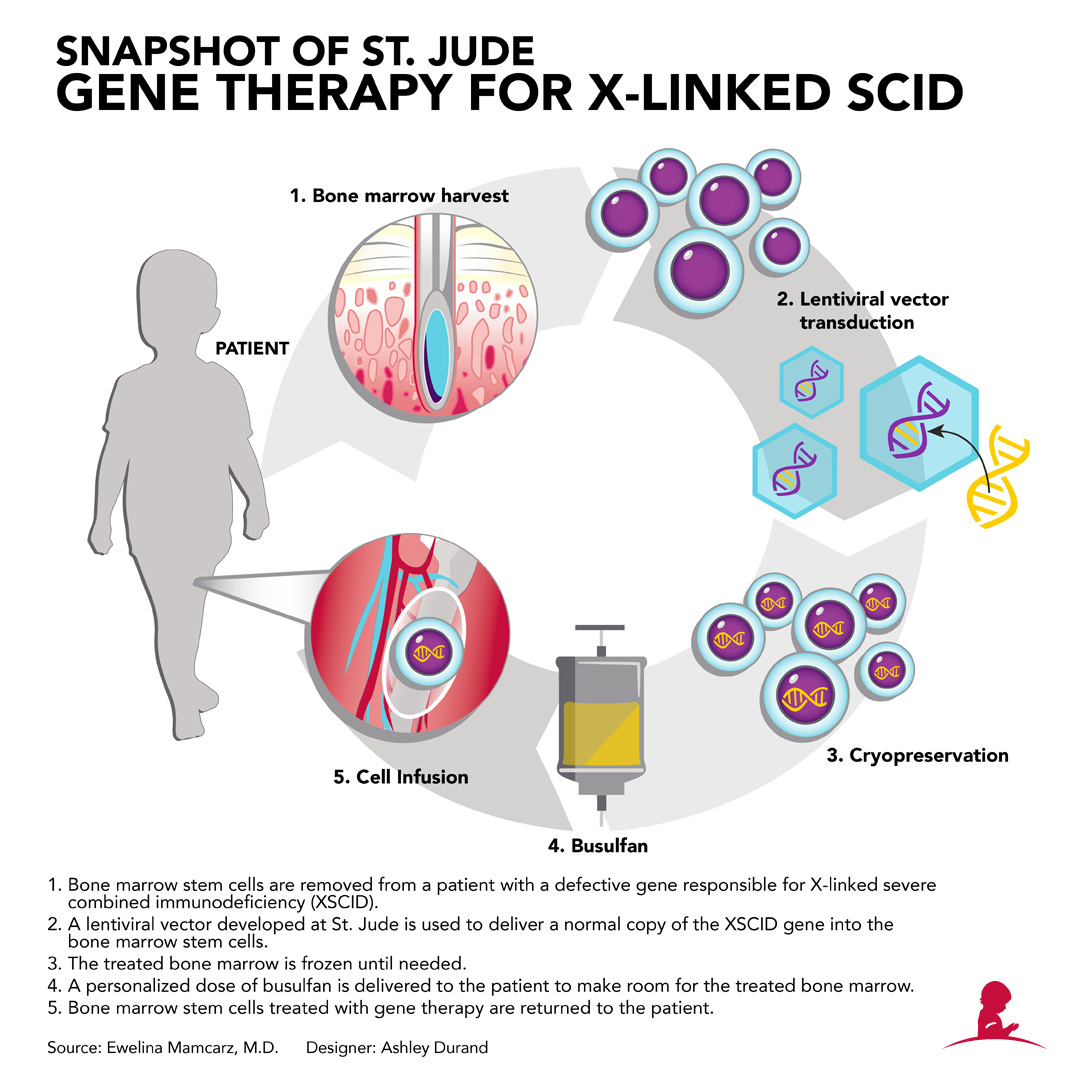
More than 150 investigational new drugs applications were filed for gene therapy in 2018 alone. Manufacturing improvements have included new chemistry, manufacturing, and controls regulations and improved accuracy of oligo synthesis. 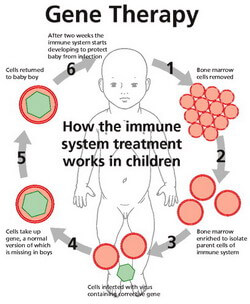
This has included a better understanding of immunogenicity and integration patterns of viral vectors as well as improved technology and modified delivery mechanisms. In the years since, better clinical and scientific understanding of the safety risks have enabled the first wave of clinical success. 2 For more, see Barbara Sibbald, “Death but one unintended consequence of gene-therapy trial,” Canadian Medical Association journal, May 2001, Volume 164, Number 11, p. The first set of promising gene therapies were brought to a halt after the 1999 death of Jesse Gelsinger from an immune reaction to the vector transporting a gene therapy for his metabolic disorder, and the development of leukemia by multiple patients-including one who died-in trials that ran between 19 for X-linked severe combined immunodeficiency (SCID-X). However, it also creates new challenges for key stakeholders-including pharma companies, regulatory agencies, providers and payers-in how to recalibrate the pharma development and reimbursement model for therapies that go beyond our traditional approach to treating disease. The bevy of new gene therapies in the development pipeline has the potential to transform care across several therapeutic areas. These successes were largely due to a better clinical and scientific understanding of safety profiles as well as a refined manufacturing process that met the consistency and quality standards required for clinical scale. Only in the past few years has that promise started to become reality: several RNA- and DNA-based therapies are now on market, and the first curative gene therapy, Luxturna, was approved in 2018. 1 International Human Genome Consortium, “Initial sequencing and analysis of the human genome,” Nature, February 2001, Volume 409, pp. The completion of the first draft of the human genome in 2001 was supposed to kick off an era of personalized medicine and curative gene therapies.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
